The term valence refers to the ability of an element to form bonds with other atoms. It is located on the outermost shell (in this case, the shell resembles a ring). This also means that the number of valence electrons that an element has determines its reactivity, electronegativity, and the number of bonds it can form.įor example, in the figure below showing a simplified diagram of sodium’s electron configuration, the valence electron is shown in red. These electrons, being the furthest from the nucleus and thus the least tightly held by the atom, are the electrons that participate in bonds and reactions. Valence electrons are electrons that located in the outermost electron shell of an atom. What are valence electrons? Why are they significant? We will also learn how to tell how many valence electrons an element has. 9.9: Periodic Trends- Atomic Size, Ionization Energy, and Metallic Character Certain properties-notably atomic radius, ionization energy, electron affinity and metallic character-can be qualitatively understood by the positions of the elements on the periodic table.In this tutorial, we learn about valence electrons, what they are, and why they are significant.9.8: The Explanatory Power of the Quantum-Mechanical Model The chemical properties of elements is determined primarily by the number and distribution of valence electrons.Electron configurations can be predicted by the position of an atom on the periodic table. 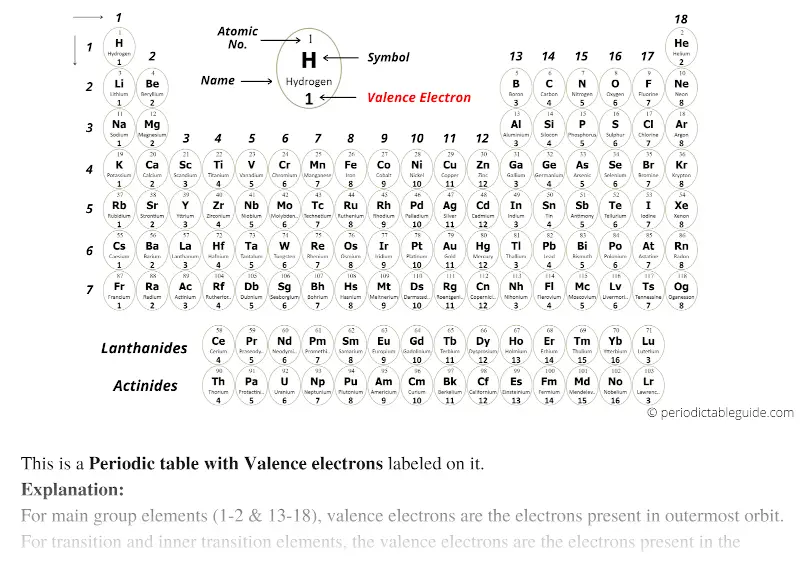
9.7: Electron Configurations and the Periodic Table The arrangement of electrons in atoms is responsible for the shape of the periodic table.Orbital filling diagrams are a way of indicating electron locations in orbitals. Hund's rule specifies the order of electron filling within a set of orbitals. The Aufbau principle gives the order of electron filling in an atom. Electron configuration notation simplifies the indication of where electrons are located in a specific atom. 9.6: Quantum-Mechanical Orbitals and Electron Configurations We look at the four quantum numbers for a given electron.Orbitals are mathematically derived regions of space with different probabilities of having an electron. This field deals with probabilities since we cannot definitely locate a particle. 9.5: The Quantum-Mechanical Model- Atoms with Orbitals Quantum mechanics involves the study of material at the atomic level.The state of electrons in atoms is described by four quantum numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
